Are you looking for chemistry chapter 4 The d and f block elements handwritten notes? you are at the right place. A good notes can boost your exam preparation.
Key Aspects
| Chapter Name | The d and f block elements |
| Chapter Number | 4 |
| Board | CBSE |
| Subject | Chemistry |
| Medium | English Medium |
Here we have shared CBSE Class 12 Chemistry Chapter 4 Notes – The d and f block elements. This will be very helpful for your exam preparation and you can score maximum marks with the help of these notes. We have covered all topics of solutions chapters. All the notes are easy and free to access.
The d and f block elements Handwritten Notes





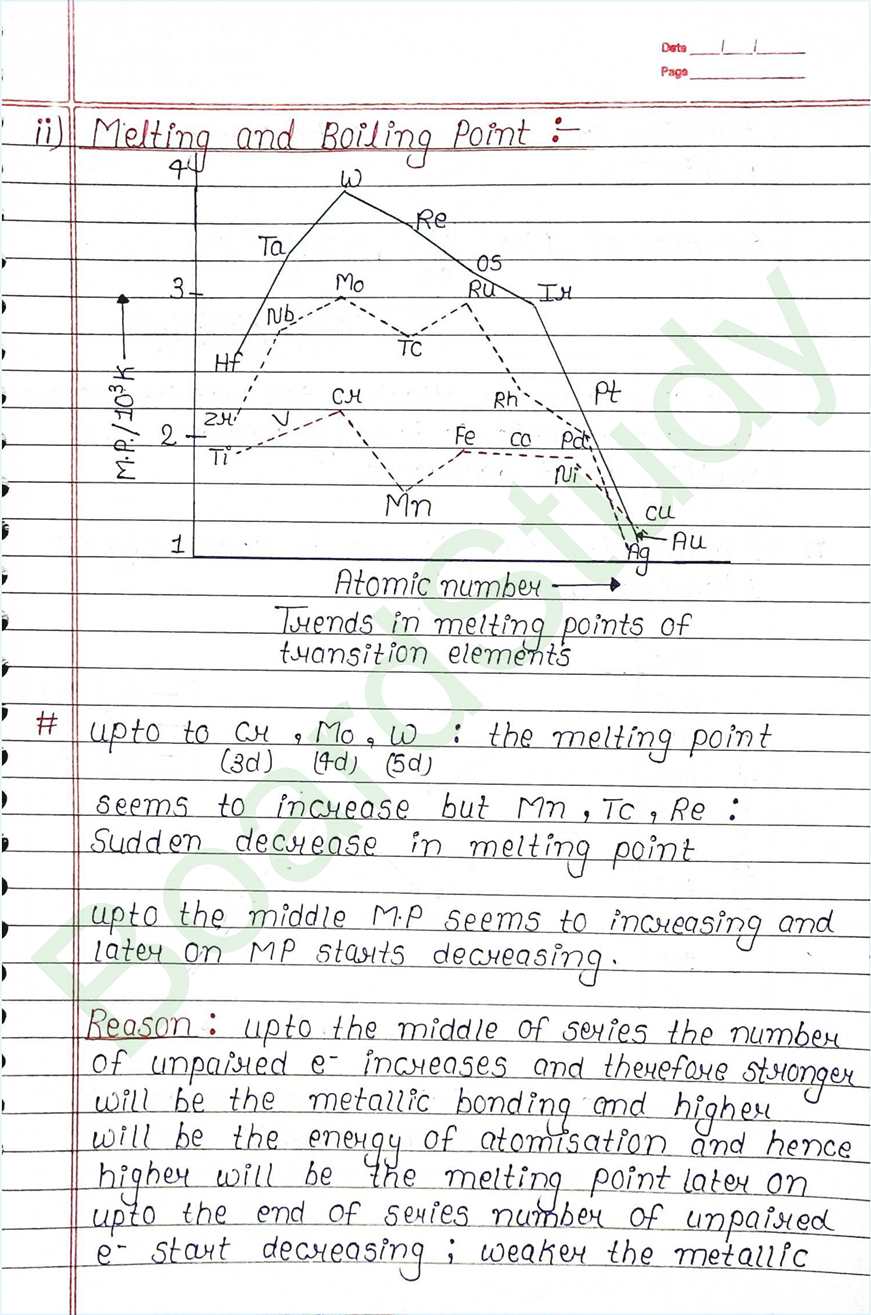
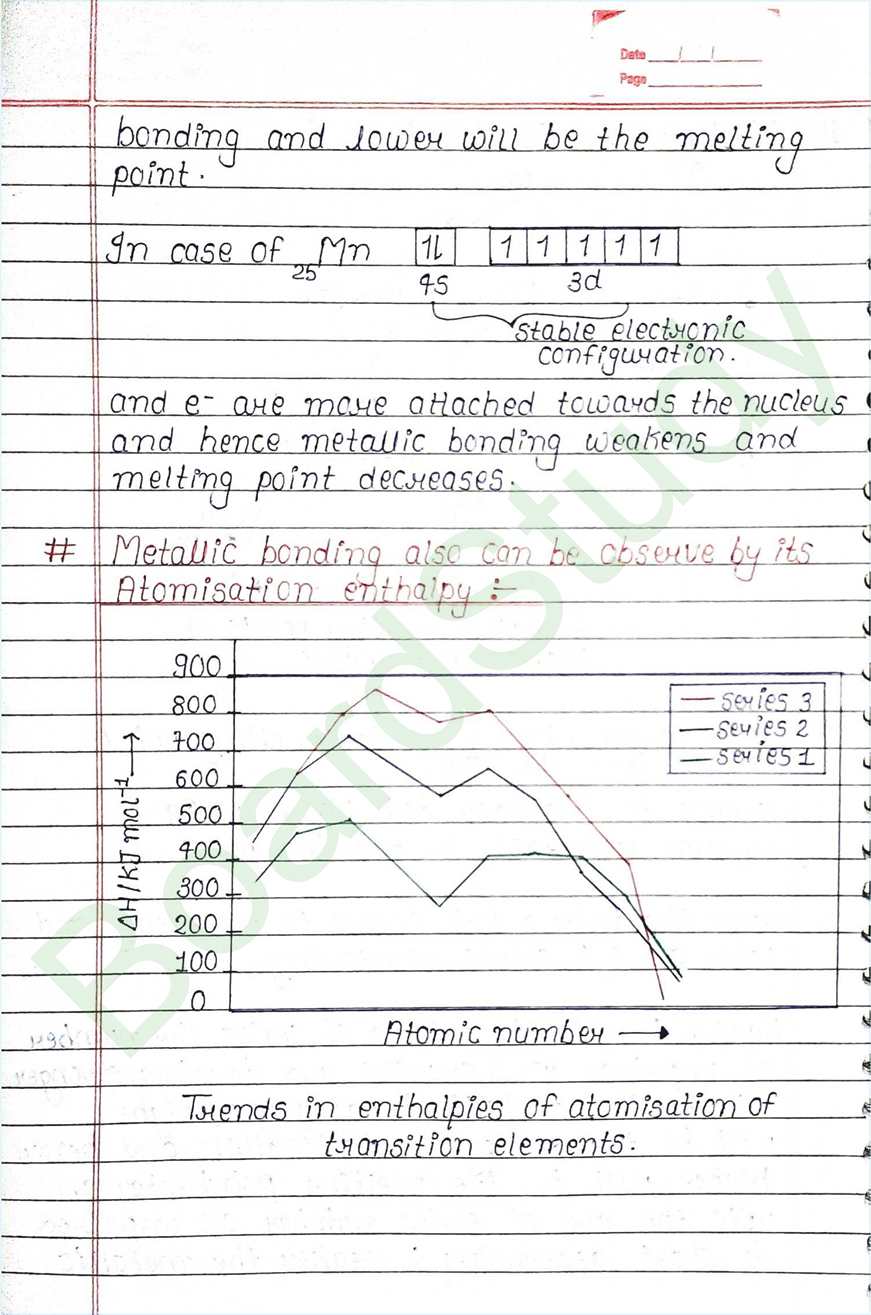
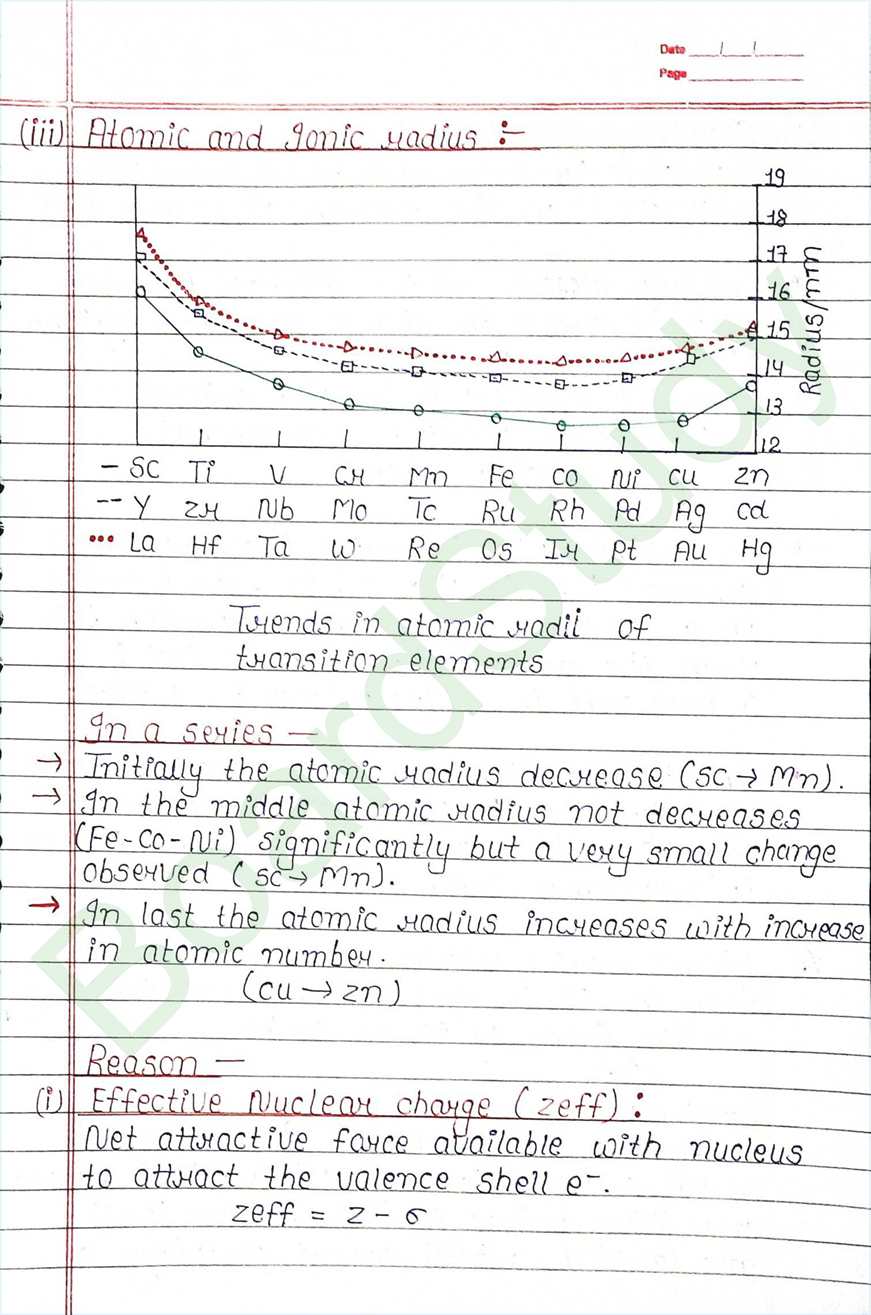






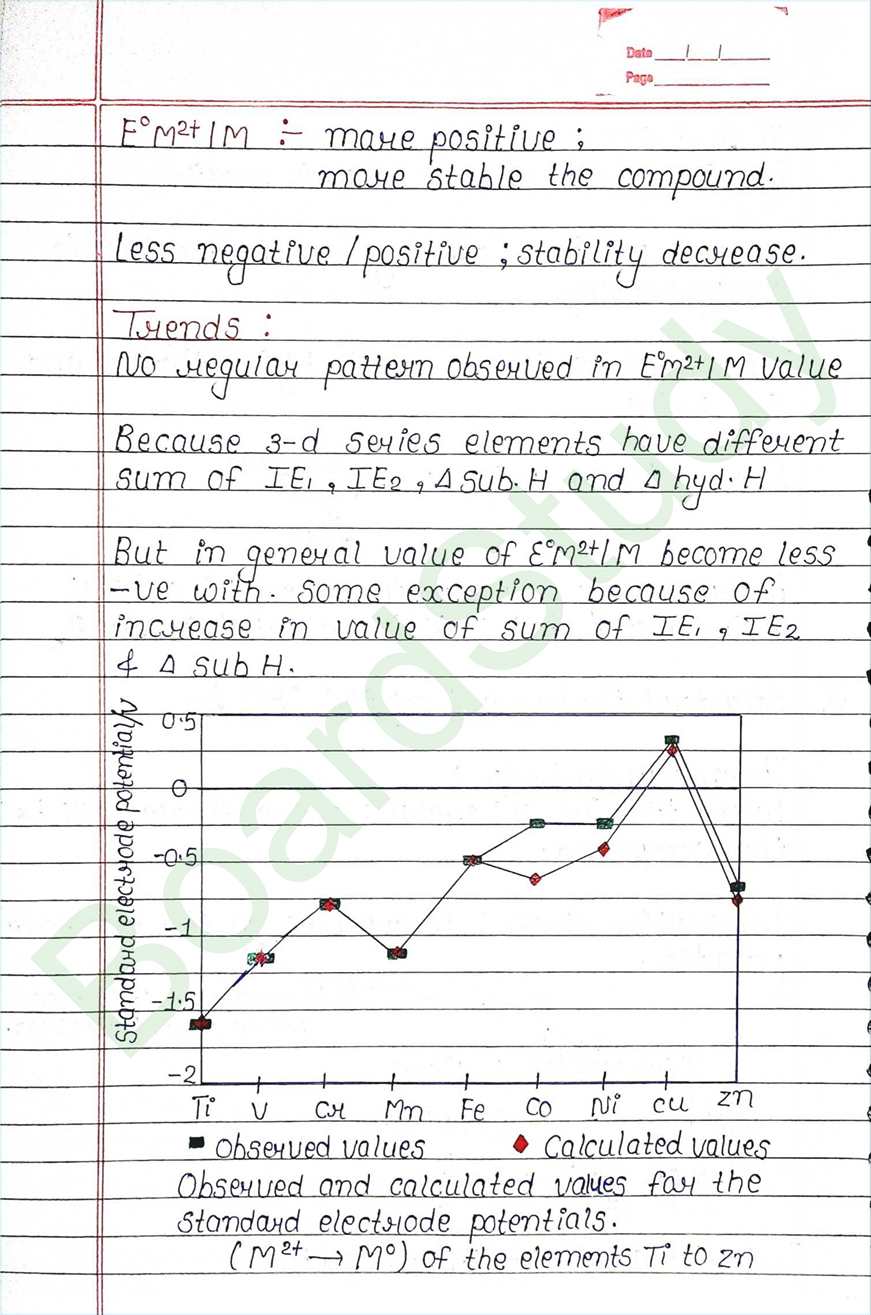






























Next Chapter: Coordination Compounds
Previous Chapter: Chemical Kinetics
Hope this notes will help you and you can help your friends by sharing this notes. Best of luck for your future exam and keep visiting the website. If you have any doubt just do comment we will be happy to help you.
Tagged with: Class 12 Chemistry Chapter 8 Notes | CLASS 12TH | d and f block | d and f block elements class 12 notes
